Ammonium chlorate Formula
Ammonium chlorate is a very unstable salt that is mainly used to manufacture explosives. Its pure form is extremely unstable and can easily explode.
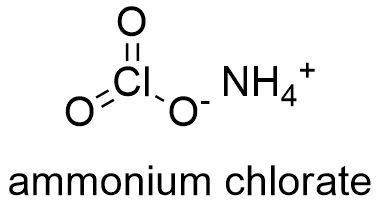
Formula and structure: The ammonium chlorate chemical formula is NH4ClO3. Its molar mass is 101. 4897 g mol-1. The ammonium chlorate structure is formed by the cation ammonium NH4+ and the anion ClO3-. The crystal structure of this salt has a rhomboidal geometry, similar to other salts such as potassium bromide. Its chemical structure can be written as below, in the common representations used for organic molecules.
Occurrence: Ammonium chlorate is very unstable, consequently it has not been found in nature.
Preparation: Ammonium chlorate is prepared from the neutralization of chloric acid with ammonia or ammonium carbonate:
HClO3 + NH3 → NH4ClO3
It can be prepared by reacting barium or calcium chlorate with ammonium sulfate to obtain the ammonium chlorate and barium or calcium sulfate. Calcium and barium cations form insoluble sulfates in water, thus it can be easily removed from the reaction mix.
Ca(ClO3)2 + (NH4)2SO4 → 2NH4ClO3 + CaSO4(s)
Physical properties: ammonium chlorate is a colorless crystal salt. Its density is 2.42 g mL-1 and its melting point is 380 ºC. However, in temperatures >142 ºC it decomposes to form nitrogen, chlorine and oxygen. It is soluble in water and diluted alcohol solutions but it is insoluble in pure alcohols.
Chemical properties: ammonium chlorate is a strong oxidizer, however, its utilization in chemical industry is very limited because its instability. When it is exposed to light can explode and even its diluted solutions are dangerous. The main reason for the unstable behavior of this salt, it is the chemical nature of every ion, ammonium cation is a potent reducer, while chlorate anion is an oxidizer.
Uses: ammonium chlorate has very limited application. Typically for the unstable salts, the main application is to manufacture explosive or chemical weapons. Ammonium chlorate
Health effects/safety hazards: ammonium chlorate is highly dangerous. Its transportation is prohibited because there is a high risk of explosions. It can violently explodes in concentrated solutions, when exposed to light or heat.
|
Related Links: |
Related Topics
Ionic and Net Ionic Equations
