Magnesium hydroxide Formula
Magnesium hydroxide, also known as milk of magnesia or magnesium dyhydroxide, is an inorganic base used as antacids and laxative.
Formula and structure: The magnesium hydroxide chemical formula is Mg(OH)2 and its molar mass is 58.3197 g mol-1. Magnesium hydroxide molecule is formed by the magnesium cation Mg+2 and the two hydroxyl groups OH-. The magnesium hydroxide lattice has a hexagonal structure. Its chemical structure can be written as below, in the common representations used for organic molecules.
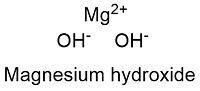
Occurrence: Magnesium hydroxide can be found in nature in the some minerals such as brucite and clay minerals.
Preparation: Magnesium hydroxide is produced by the reaction of magnesium salts as carbonate and chloride and sodium hydroxide. It is reaction can occur due the solubility product (Kps) of magnesium hydroxide is low.
Mg +2 + 2 OH- → Mg(OH)2
Physical properties: Magnesium hydroxide is white, odorless solid. Its density is 2.346 g mL-1. The melting point is 350 ºC and above this temperature, the magnesium hydroxide decomposes. It is poorly soluble in water, ethanol and methanol.
Chemical properties: Magnesium hydroxide is the main component of milk of magnesium. It can be used as antacids because the basicity of the hydroxides neutralize the H+ present in the stomach acid (HCl). Moreover, the laxative effect of milk of magnesium is due the hydroxide ions can retain molecules water (through hydrogen bonds) in its way through the intestines, promoting the intestinal movement.
Uses: The most known application of magnesium hydroxide is as milk of magnesia, however, it has many uses such as being a component of electrical and thermal conductor. Magnesium hydroxide is also used in the waste water treatment and as fire retardant.
Health effects / safety hazards: Magnesium hydroxide may cause eye irritation. Magnesium hydroxide is inert: it is not flammable and it does not react with other chemical compounds at room temperature. It cannot be consume in excess because may cause dehydratation.
|
Related Links: |
